So for those of you that have read my 'garage build' thread you will have seen that I've been building in a lab area and I thought it was probably an good idea to explain what I was using it for as it's not really the kind of thing 'car people' normally stick in the back of their garage..
It all started about 5 or 6 years ago. Myself and a few of my friends have been into various 'treasure hunting' hobbies for years (think metal detecting, bottle digging etc.) Around 2016 however we started heavily focusing on what we could find in the local rivers. ('Mudlarking' is the term some people use but there's not much mud in the brick bottomed rivers of Birmingham!) Anyway, we had found a number of items, mainly coins and jewelry that we suspected to be gold or silver but had no real idea how to test them. I started off by simply googling some info on what different acids etc would do to the various precious metals so I could work out what we had but before I knew it I was onto reading about metal refining and this all happened..
At first I started out just doing some experiments under the extraction in my spray booth but that proved not to be a particularly good idea as the fumes generated doing this are not only toxic but extremely corrosive too. I ended up building a makeshift lab in the old ladies toilet at the back of my workshop which I used for a number of years until I started to think about replacing it with an all singing all dancing professional setup.
What really got me hooked was working out some of the more obscure places these metals can be found and quite how valuable a small amount can be.. I had an idea that things like electronic components or catalytic converters contained precious metals but don't think I really had a grasp of quite how much value was out there lurking. (this was pre cats being worth a fortune era!) Also being into exploring old derelict buildings. I've seen so many rooms stacked to the ceiling with old computers rotting away and just walked on by thinking it was all worthless but in reality there's value in all sorts of items from old x-ray film to road dirt! It's just coming up with a way to extract it economically...
So I could write a whole book on this stuff (and people have) but i thought I'd put up a few photos of some of the more basic work and if people find it interesting I can maybe go into more detail.
Refining can usually be achieved in one of three ways, either by dissolution and precipitation, smelting and cupulation, or by electrolysis. Each method has its pros and cons but in this case we're going to be looking dissolving and precipitating some gold from old ceramic CPUs as this is fairly simple
Refining gold is actually quite easy compared to the other metals. The main problem is gold is very unreactive. Nearly all acids wont touch it and mercury and cyanide (that will) pose some obvious health hazards. Luckily for us, someone a long time ago realized that although hydrochloric and nitric acid on their own won't dissolve the gold a mixture of the two will. Therefore the first step is to cover the metals we want to refine with hydrochloric acid and heat the solution. Once hot we can add small doses of nitric acid and the reaction starts. The nitric acid oxides the gold and the gold oxide is able to react with the HCl and dissolve into solution as a satisfyingly gold coloured Auric Chloride.
Most 'impurities' will dissolve too. You've got to have some fairly exotic metals in there to resist this 'aqua regia' solution as it is known. In the case of these CPUs they are mostly inert ceramic but the pins are made from an alloy known as 'kovar' which is mainly iron and nickel. Luckily these don't really cause us any problems. They simply dissolve into the solution along with the gold plating. Also it's not the obvious plating that we are really after with these . The majority of the gold is present in the brazing that is used to solder the ceramic integrated circuit to the ceramic body. This brazing is mainly an alloy of gold and silver, the gold will dissolve but the silver forms silver chloride which is insoluble and hangs around int he solution making it look very cloudy. The only other problem metal is tungsten which is present in some of the ceramic heat spreaders. This batch didn't have many of the type of cpus that have these spreaders but there was a few in there and the tungsten reacted to form a really bright yellow tungsten oxide which also hangs around undissolved and makes everything a powdery yellow..
Once all the metals are dissolved any excess of nitric acid must be eliminated. There is a number of methods of achieving that but I prefer to add a small amount of sulphamic acid. Also at this point it is good practice to add a few drops of sulphuric acid to precipitate out any lead that may have dissolved as a solid lead sulphate. The solution is filtered which should separate all these undissolved contaminates and just leave us with the liquids.
We are now left with a solution containing our gold but also lots of iron, nickle and probably a bit of copper and even some bonus platinum. Luckily there is a way to selectively precipitate out the gold using sulpher dioxide gas. This can be done by literally bubbling the gas through the solution but it can also be done more simply by adding sodium metabisulphite powder which reacts with the residual acid and creates the dioxide gas directly in the solution. For reasons i don't quite understand this gas pretty much only precipitates the gold. Even the platinum which really doesn't like being in solution stays put allowing almost metallic gold to collect on the bottom of the beaker.
The last step is another filtration to catch the gold powder and a repeat of the process to ensure all the various contaminates are removed before the gold can be melted and if everything is don't right is essentially 24k 999 pure. There are some stumbling blocks but overall its quite simple once you've done it a few times.

Starting material

Partial crushing to speed the process up a bit. I'm mainly aiming to break up the silicone dies and expose the braze joining them to the ceramic.

Cover with Hydrochloric acid at around 30% concentration

Heat and start adding small doses of Nitric acid.

Reaction gets going and metals start to go into solution turning it dark. The orange/brown gas given off is Nitrogen Dioxide from the Nitric acid decomposing. If it helps you visualize the smell of this is what the science labs at school used to smell like! (not that you want to be smelling it too much, its very toxic)

It usually takes about a day on the heat to dissolve all the metals in these and i do two extractions on each batch.

Next the solutions are filtered to remove the insoluble stuff.

Nearly all metal has gone into solution
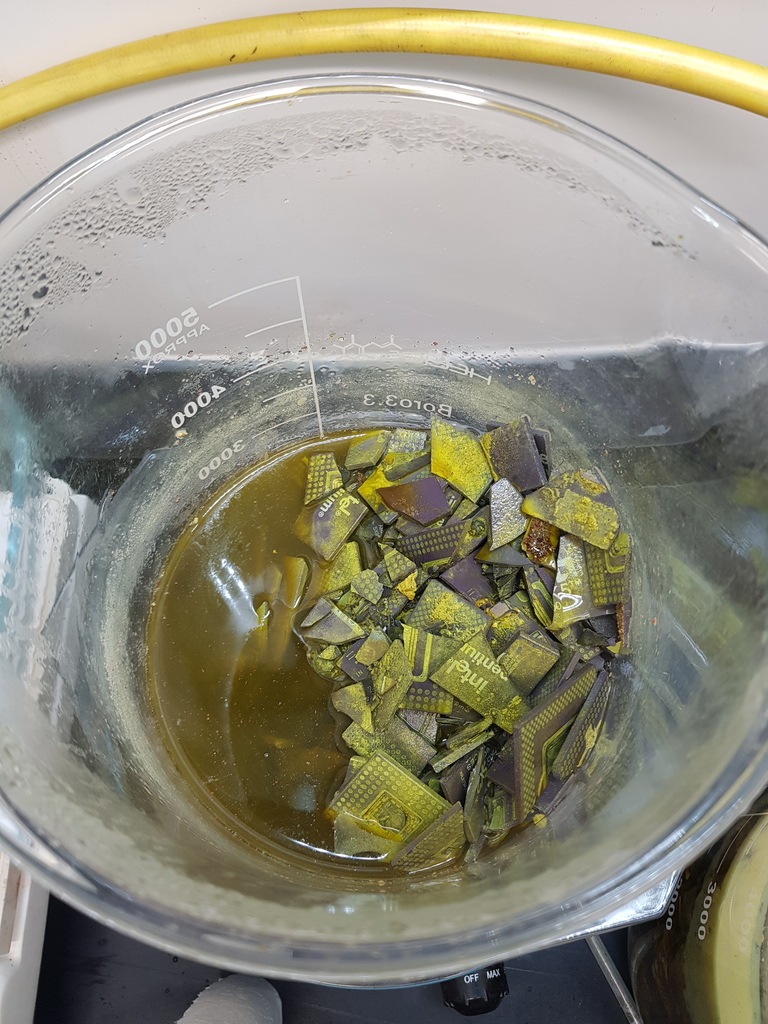
You can see some of the yellow Tungsten Oxide and the Silver Chloride forming a scum on the beaker.

The Sodium Metabisulphate is then added to the filtered liquid and we filter that again to get out gold powder.

Then i redissolve it again as the first part of the process probably lets some small amounts of impurities through. This time I'm using a more elaborate setup which helps reduce the loss of acid vapors and catches some of NOx gas which can be recycled into new nitric acid. Its debatable if it is worth it but it puts a lot less nasties into the atmosphere doing it this way so i make an effot!

Reaction staring up again. The filter paper will just break up in the hot acid.

It also helps that you can stir with a magnetic stir bar when there not a load of ceramics in the way.

Dissolution complete, much cleaner looking, probably 99% gold chloride.

Final filter

After dropping the gold out of the solution again with SMB

Time to melt

Just quickly melted using oxy acetylene torch. The filter paper just burns away.

Our final product. 999 fine gold, about £1500 worth.
[/font]
It all started about 5 or 6 years ago. Myself and a few of my friends have been into various 'treasure hunting' hobbies for years (think metal detecting, bottle digging etc.) Around 2016 however we started heavily focusing on what we could find in the local rivers. ('Mudlarking' is the term some people use but there's not much mud in the brick bottomed rivers of Birmingham!) Anyway, we had found a number of items, mainly coins and jewelry that we suspected to be gold or silver but had no real idea how to test them. I started off by simply googling some info on what different acids etc would do to the various precious metals so I could work out what we had but before I knew it I was onto reading about metal refining and this all happened..
At first I started out just doing some experiments under the extraction in my spray booth but that proved not to be a particularly good idea as the fumes generated doing this are not only toxic but extremely corrosive too. I ended up building a makeshift lab in the old ladies toilet at the back of my workshop which I used for a number of years until I started to think about replacing it with an all singing all dancing professional setup.
What really got me hooked was working out some of the more obscure places these metals can be found and quite how valuable a small amount can be.. I had an idea that things like electronic components or catalytic converters contained precious metals but don't think I really had a grasp of quite how much value was out there lurking. (this was pre cats being worth a fortune era!) Also being into exploring old derelict buildings. I've seen so many rooms stacked to the ceiling with old computers rotting away and just walked on by thinking it was all worthless but in reality there's value in all sorts of items from old x-ray film to road dirt! It's just coming up with a way to extract it economically...
So I could write a whole book on this stuff (and people have) but i thought I'd put up a few photos of some of the more basic work and if people find it interesting I can maybe go into more detail.
Refining can usually be achieved in one of three ways, either by dissolution and precipitation, smelting and cupulation, or by electrolysis. Each method has its pros and cons but in this case we're going to be looking dissolving and precipitating some gold from old ceramic CPUs as this is fairly simple
Refining gold is actually quite easy compared to the other metals. The main problem is gold is very unreactive. Nearly all acids wont touch it and mercury and cyanide (that will) pose some obvious health hazards. Luckily for us, someone a long time ago realized that although hydrochloric and nitric acid on their own won't dissolve the gold a mixture of the two will. Therefore the first step is to cover the metals we want to refine with hydrochloric acid and heat the solution. Once hot we can add small doses of nitric acid and the reaction starts. The nitric acid oxides the gold and the gold oxide is able to react with the HCl and dissolve into solution as a satisfyingly gold coloured Auric Chloride.
Most 'impurities' will dissolve too. You've got to have some fairly exotic metals in there to resist this 'aqua regia' solution as it is known. In the case of these CPUs they are mostly inert ceramic but the pins are made from an alloy known as 'kovar' which is mainly iron and nickel. Luckily these don't really cause us any problems. They simply dissolve into the solution along with the gold plating. Also it's not the obvious plating that we are really after with these . The majority of the gold is present in the brazing that is used to solder the ceramic integrated circuit to the ceramic body. This brazing is mainly an alloy of gold and silver, the gold will dissolve but the silver forms silver chloride which is insoluble and hangs around int he solution making it look very cloudy. The only other problem metal is tungsten which is present in some of the ceramic heat spreaders. This batch didn't have many of the type of cpus that have these spreaders but there was a few in there and the tungsten reacted to form a really bright yellow tungsten oxide which also hangs around undissolved and makes everything a powdery yellow..
Once all the metals are dissolved any excess of nitric acid must be eliminated. There is a number of methods of achieving that but I prefer to add a small amount of sulphamic acid. Also at this point it is good practice to add a few drops of sulphuric acid to precipitate out any lead that may have dissolved as a solid lead sulphate. The solution is filtered which should separate all these undissolved contaminates and just leave us with the liquids.
We are now left with a solution containing our gold but also lots of iron, nickle and probably a bit of copper and even some bonus platinum. Luckily there is a way to selectively precipitate out the gold using sulpher dioxide gas. This can be done by literally bubbling the gas through the solution but it can also be done more simply by adding sodium metabisulphite powder which reacts with the residual acid and creates the dioxide gas directly in the solution. For reasons i don't quite understand this gas pretty much only precipitates the gold. Even the platinum which really doesn't like being in solution stays put allowing almost metallic gold to collect on the bottom of the beaker.
The last step is another filtration to catch the gold powder and a repeat of the process to ensure all the various contaminates are removed before the gold can be melted and if everything is don't right is essentially 24k 999 pure. There are some stumbling blocks but overall its quite simple once you've done it a few times.

Starting material

Partial crushing to speed the process up a bit. I'm mainly aiming to break up the silicone dies and expose the braze joining them to the ceramic.

Cover with Hydrochloric acid at around 30% concentration

Heat and start adding small doses of Nitric acid.

Reaction gets going and metals start to go into solution turning it dark. The orange/brown gas given off is Nitrogen Dioxide from the Nitric acid decomposing. If it helps you visualize the smell of this is what the science labs at school used to smell like! (not that you want to be smelling it too much, its very toxic)

It usually takes about a day on the heat to dissolve all the metals in these and i do two extractions on each batch.

Next the solutions are filtered to remove the insoluble stuff.

Nearly all metal has gone into solution
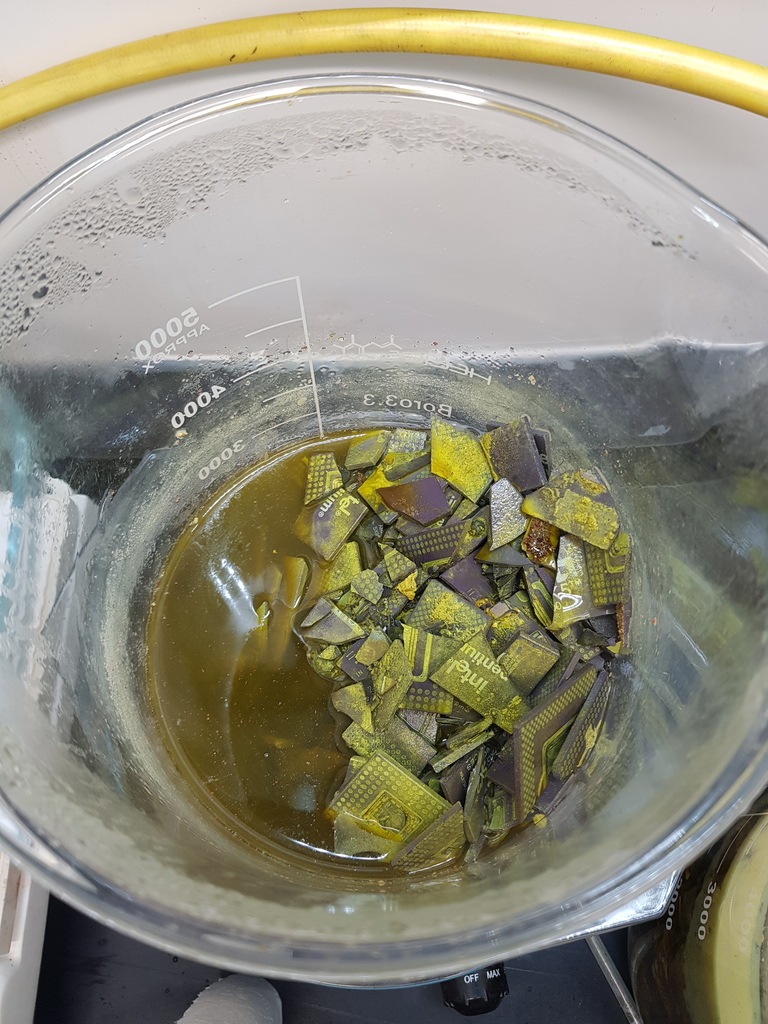
You can see some of the yellow Tungsten Oxide and the Silver Chloride forming a scum on the beaker.

The Sodium Metabisulphate is then added to the filtered liquid and we filter that again to get out gold powder.

Then i redissolve it again as the first part of the process probably lets some small amounts of impurities through. This time I'm using a more elaborate setup which helps reduce the loss of acid vapors and catches some of NOx gas which can be recycled into new nitric acid. Its debatable if it is worth it but it puts a lot less nasties into the atmosphere doing it this way so i make an effot!

Reaction staring up again. The filter paper will just break up in the hot acid.

It also helps that you can stir with a magnetic stir bar when there not a load of ceramics in the way.

Dissolution complete, much cleaner looking, probably 99% gold chloride.

Final filter

After dropping the gold out of the solution again with SMB

Time to melt

Just quickly melted using oxy acetylene torch. The filter paper just burns away.

Our final product. 999 fine gold, about £1500 worth.
Anyway that will do for now.. If people find it interesting maybe i will get some photos of some of the other processes. I have done quite a bit of silver which has to be refined in a electrolytic cell to get it to 999 purity. I have also been experimenting with refining platinum and palladium which is tricky but quite car relevant as the main source is catalytic converters. Then theres all the various processes to automate processing different types of scrap in the first place. These ceramic cpus are by far the simplest as they don't need any kind of pre-processing other then crushing. Most other sources are not that simple else id be a millionaire!
Also excuse me if i also mention if anyone has anything to sell like jewelry, silver, scrap cats, old electronics stuff etc etc. please give me a shout. The hardest part is getting the stuff to refine in the first place so I'm more than happy to pay people what stuff is worth just for the fun of learning tbh!
Also excuse me if i also mention if anyone has anything to sell like jewelry, silver, scrap cats, old electronics stuff etc etc. please give me a shout. The hardest part is getting the stuff to refine in the first place so I'm more than happy to pay people what stuff is worth just for the fun of learning tbh!
[/font]














